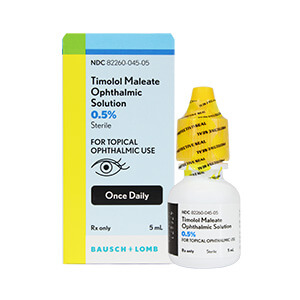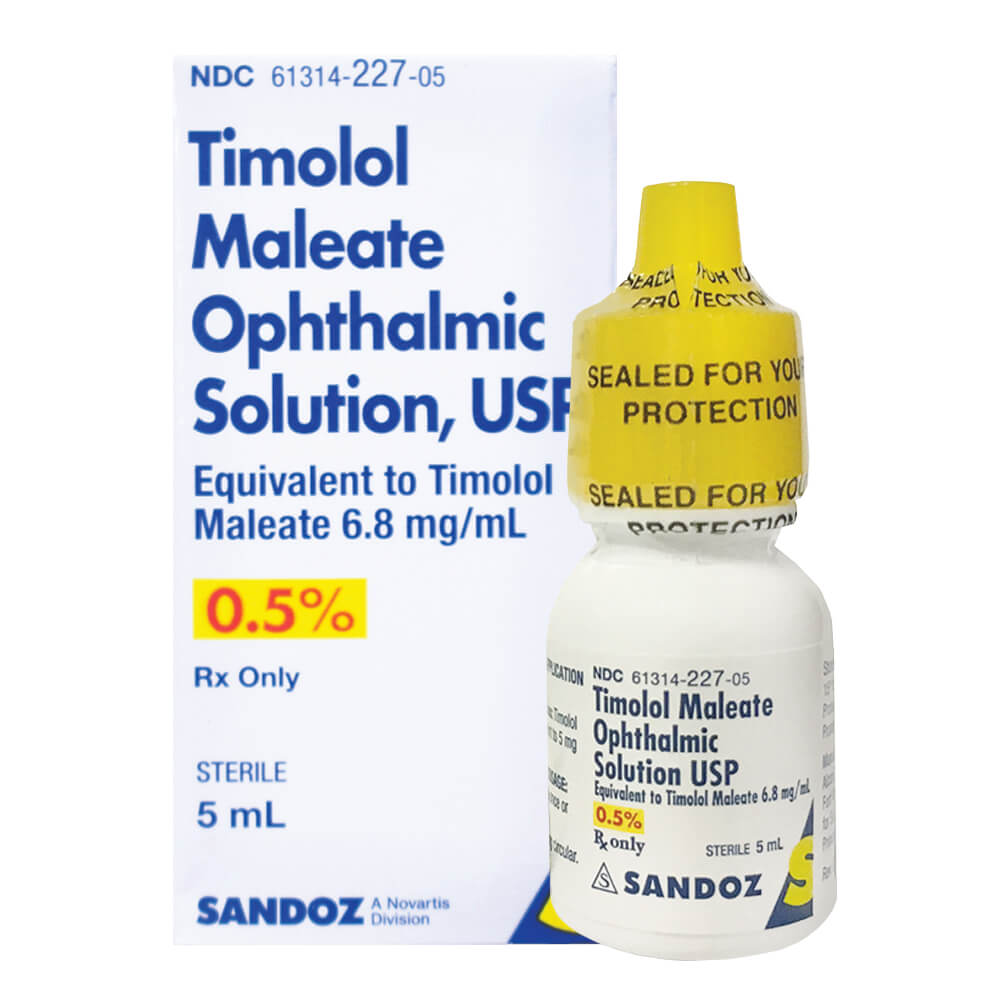
Sandoz Timolol Maleate Ophthalmic Solution 0.5% - 5mL
Sandoz Timolol Maleate Ophthalmic Solution 0.5% is used to treat elevated intraocular pressure caused by open-angle glaucoma or ocular hypertension.- Active Ingredient: 6.8 mg Timolol Maleate, a beta-blocker that reduces eye pressure
- Preservative: Benzalkonium Chloride 0.01%
- Helps prevent optic nerve damage by reducing high intraocular pressure associated with glaucoma and ocular hypertension
- Store at controlled room temperatures between 15°C-30°C (59°F-86°F)
- 5mL bottle, convenient for daily use in managing eye pressure
Compare to: Timoptic, Betimol, Istalol
Brand: Sandoz
PO-6463
This product is DSCSA tracked.
Before an order for diagnostic or pharmaceutical products can be processed AMCON is required to keep on file a copy of the Practitioner's therapeutic license. Practitioners are solely responsible for complying by the laws of their state. AMCON assumes no liability for practitioner compliance.
We cannot accept returns on pharmaceutical products.
 This product can only be shipped within the United States.
This product can only be shipped within the United States. Already an Amcon Customer?
Let's get you taken care of.
Do you have access to order on our Web Store/JAOS?
- If you have access to the Amcon Web Store, visit our web store, click the link in the top right to sign in, and purchase these products today.
- If you have access, sign in to JAOS here. If these products are not on your order form, contact your sales representative.
Don't have web access but have a customer number with Amcon Labs?
- Contact our sales team today, and we will take care of you as soon as possible!